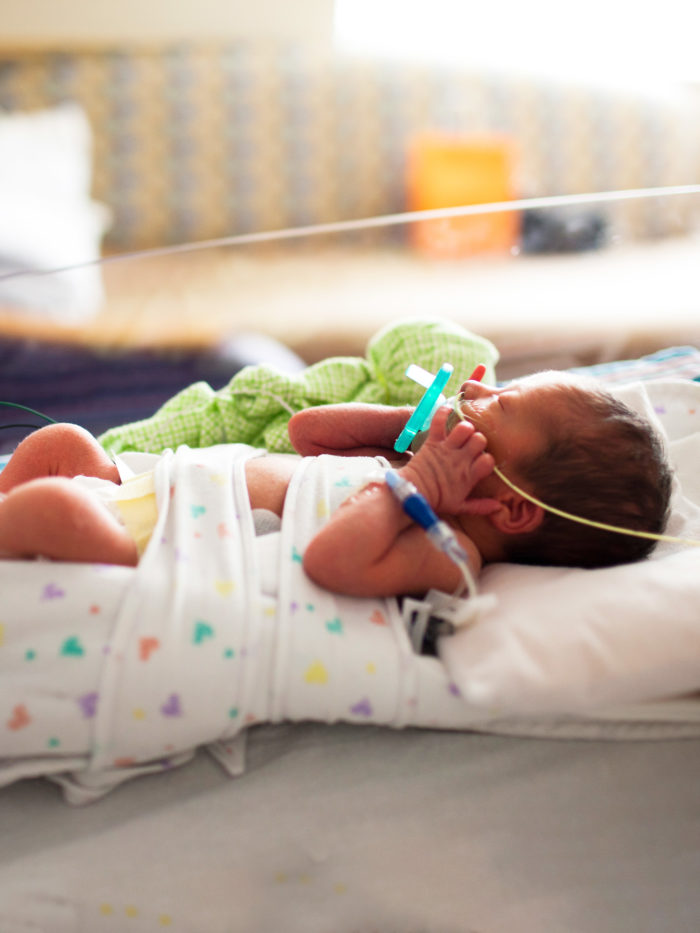
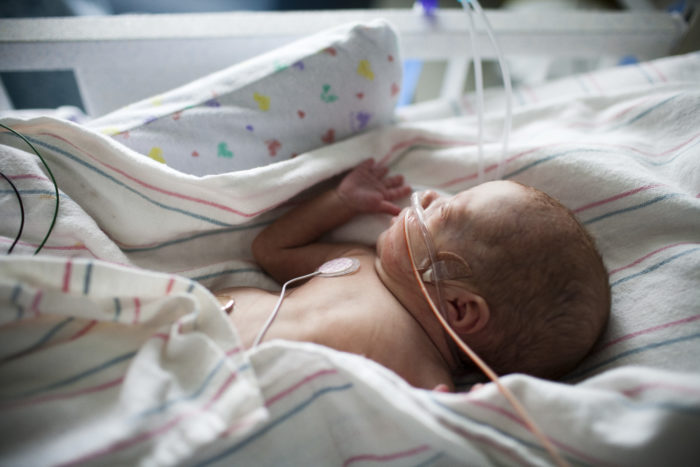
A partnership between CHOC and Masimo has developed technology to noninvasively monitor blood oxygen and hemoglobin levels in newborns.
Rapid Whole Genome Sequencing continues to provide answers and hope for parents of critically ill children with rare diseases
CHOC has ordered cutting-edge tests of rapid whole-genome sequencing (rWGS) on 150 patients, with 76 of them getting a precise diagnosis.
Rapid genome testing for infants saves lives, costs
A recently completed pilot program at CHOC and other California clinical sites helped target a baby’s specific genetic disease in just days.
CHOC to host NeoHeart: Cardiovascular Management of the Neonate Conference, March 27-29
To help address the needs of neonates and their families, CHOC will again host “NeoHeart: Cardiovascular Management of the Neonate.”